This system features an XPS system interfaced wih a 3-electrode electrochemical cell. The XPS system features a dual anode xray source, 140 mm mean radius hemispherical analyzer with channelplate detector, ion sputter gun, and sample heating and cooling. The sample is transferred from the XPS/analysis chamber to the evacuated electrochemical chamber, which is then separated from the rest of the system by a gate valve. The electrochemical chamber is backfilled to atmospheric pressure with ultrapure N2, and the electrochemical cell is admitted to the chamber so that the meniscus wets the sample surface. This permits typical electrochemical measurements (cyclic voltammetry, chronoamperometry, linear polarization, etc.). Following sample emersion at controlled potential , and rinsing in dilute electrolyte, the sample can be transferred back to UHV for further analysis.
Representative results are shown in Figure 2 (J.Liu, et al. J. Electrochem. Soc. 152 (2005) G115). An atomically clean Ru electrode surface is coated with a monolayer of iodine in UHV, then transferred to the electrochemical cell, and exposed to water vapor and to aqueous solution. The data show that a monolayer inhibits oxidation of the Ru surface even under high vapor pressure or liquid conditions—an important finding with applications in microelectronics fabrication and other areas.

Figure 1. Schematic of UHV XPS-electrochemistry system, permitting sample transfer between UHV and aqueous environments under controlled conditions.
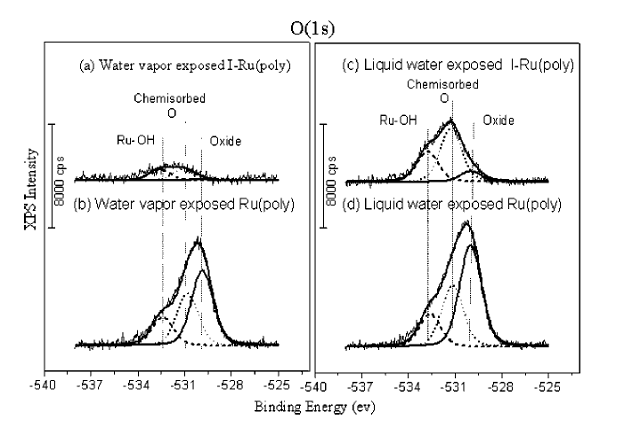
Figure 2. XPS spectra acquired in the UHV-electrochemistry system. An atomically clean Ru electrode surface is modified with a monolayer of chemisorbed iodine. The I-modified Ru electrode is exposed to water vapor (top, left) or aqueous solution (top, right) and transferred back to UHV for XPS analysis. The daa show chemisorption of OH and O, but no true oxide formation. In contrast, unmodified Ru electrodes exposed to these environments exhibit extensive RuO2 formation (left and right, bottom trace). See J. Liu, et al., J. Electrochem. Soc. 152 (2005), G115.